Technology
Fluctuation-based super-resolution traction force microscopy (FBSR-TFM) is an imaging and analysis method that enhances the spatial resolution and accuracy of conventional traction force microscopy (TFM). It allows for precise mapping of the mechanical forces that cells exert on their extracellular environment.
The key aspects of FBSR-TFM are:
-
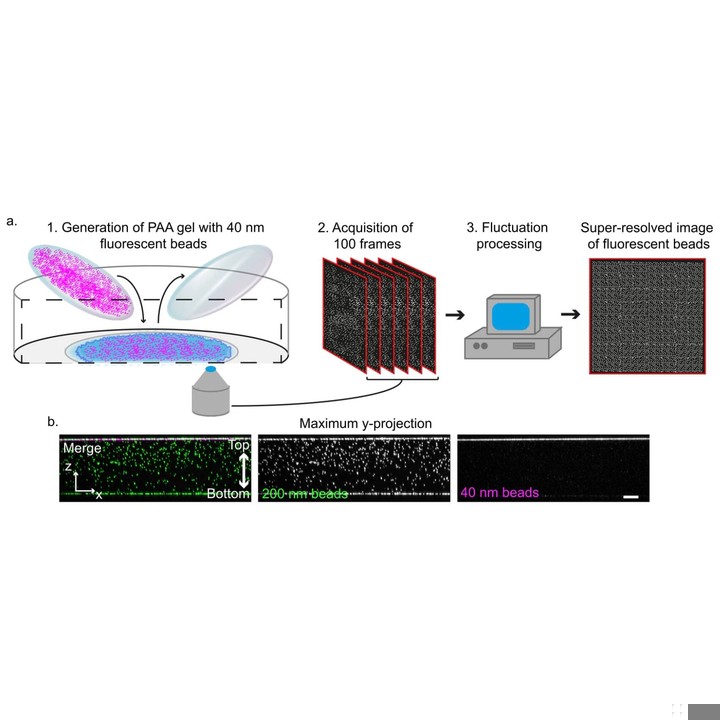
Sample preparation - Cells are plated on polyacrylamide gels containing a high density of small (40nm) fluorescent beads only on the top surface of the gel. This increased bead density compared to typical TFM gels enables improved force mapping resolution.
-
Image acquisition - Using standard fluorescence microscopes, multiple images (typically 100) are captured over time of the fluorescent beads embedded in the gel. This allows fluorescence intensity fluctuations of the beads to be measured.
-
Fluctuation-based super-resolution (FBSR) image reconstruction - The fluorescence fluctuation of the beads over time contains information about the nanoscale localization of the beads at a higher resolution than the diffraction limit. Computational algorithms like SRRF and SACD are used to perform an FBSR reconstruction to predict bead positions at super resolution, around 2-3 fold higher than the original images.
-
FBSR-TFM computation - The standard TFM computational workflow is then applied to the FBSR images. This involves: image alignment of the beads before and after cell detachment, tracking bead displacements caused by cell forces, and calculating cellular traction forces and maps from the measured displacements using mathematical frameworks like FTTC.
-
Enhanced TFM outputs - By increasing the density of analyzed beads 5-10 fold using FBSR and improving bead tracking accuracy, FBSR-TFM significantly enhances the spatial resolution and quality of measured cellular traction forces, displacements, and maps compared to standard TFM. Force hotspots corresponding to focal adhesions are clearly resolved.
Key benefits of FBSR-TFM:
- Simplicity - Uses standard sample prep and imaging systems, only adding FBSR computational reconstruction
- Compatibility - Works seamlessly with existing open-source TFM software
- Multiplexing - Enables simultaneous high-resolution imaging of cell structures like focal adhesions
- Live cell - Capable of fast imaging for measuring real-time traction force dynamics
- Resolution - Resolves traction forces with higher precision and detail than standard TFM
- Biological insight - Reveals new biomechanical relationships, e.g between focal adhesions and filopodia
FBSR-TFM significantly pushes the capabilities of TFM by enhancing resolution, detail, and accuracy of measured cellular forces through increased analyzed bead density and tracking precision enabled by fluorescence fluctuation super-resolution imaging. This allows new biological insights into sub-cellular force generation and transmission.
Publications featuring FBSR-TFM
__wordcloud.jpg)
|
Fluctuation-based super-resolution traction force microscopy Aki Stubb, Romain F Laine, Mitro Miihkinen, Hellyeh Hamidi, Camilo Guzmán, Ricardo Henriques, Guillaume Jacquemet, Johanna Ivaska Paper published in Nano letters, January 2020 Technologies: FBSR-TFM, NanoJ (), NanoJ-SQUIRREL () and NanoJ-SRRF () Funded by: BBSRC DOI: 10.1021/acs.nanolett.9b04083 |
|
Funding contributing to FBSR-TFM

|
Unveiling live-cell viral replication at the nanoscale Ricardo Henriques Funded by: EMBO - Installation Grant Duration: January 2021 - December 2025 Publications: 34 |